
Polyethylene terephthalate (PET) is a low diffusive polymer compared to other packaging polymer like polyolefins, which means that the migration of substances from the polymer into food is low. As a consequence the migration of the residual monomers ethylene glycol and terephthalic acid or the intentionally added polymerisation catalyst antimony is typically far below of the specific migration limits (for a literature compilation see Welle 2014).
PET is therefore a perfect material for food packaging. On the other hand the migration of acetaldehyde from PET beverage bottles is sometimes responsible for consumer complaints e.g. off-flavors in PET packed natural mineral water. Acetaldehyde is a degradation product of PET and can be considered as the most important non-intentionally added substance (NIAS) in PET. Other NIAS found in PET are for example impurities or oxidation products of recyclates, colorants and masterbatches like solvents or aldehydes. Also reaction products of oxygen scavengers can be considered as NIAS when these active additives are applied to PET bottles. Within the last few years, NIAS are getting more and more important in food law compliances evaluation of food packaging materials. However, clear strategies for the food law compliance evaluation and analytical testing methods are not available until now.

Acetaldehyde
10 µg/l is typically applied. Potential genotoxic compounds should be minimized in their concentration to a non-detectable level in the PET bottles. However, a migration threshold limit of 0.15 µg/l can be applied for genotoxic substances. These concentrations are – compared to other specific migration limits (SML) – extremely low, which are from an analytical point of view very challenging. Especially due to the fact that NIAS are in the nature of things non-intentionally added. Therefore, it is not completely known which NIAS occur in PET bottles in day by day production which is a risk for preform manufacturers as well as bottling companies.
Anthranilamide
In order to control the amounts of NIAS in PET preforms and bottles and to minimize the risk of complaints, analytical screening methods need to be applied. Non-target analytical screening methods with detection limits of 10 µg/l (or lower) are very labor-intensive and expensive, because the detection limits of such non-target screening methods should be in minimum a factor of ten below of the specific migration limit. In addition, the migration is a time consuming process. Storage times of ten days in minimum are typically applied. For production control of PET preforms or bottles such long storage times are not useful.
Terephthalic acid
On the first glance NIAS in PET bottles seem to be a new topic for the PET beverage business. The evaluation of post-consumer recyclates, which has been successfully practiced over the last two decades, however, is nothing else than an evaluation of substances which are non-intentionally added to the packaging material. Huge experiences have been collected in the analysis and evaluation of post-consumer (non-intentionally added) substances at the Fraunhofer IVV. Fast and validated screening methods have been developed for the screening of any unwanted substances occurring in PET preforms and bottles. On the second glance the food law compliance evaluation is much harder than NIAS evaluation in PET preform production, because single, recollected post-consumer bottles might be contaminated e.g. if the bottle were misused for storage of garden chemicals, paints or solvents. A 100% control of each post-consumer bottle foreseen for recycling is impossible. In preform production NIAS occur most likely in the whole production batch or at least over a certain production period.
Bisphenol A
The safety of post-consumer recycled PET is evaluated with a combination of material tests on the PET material and migration modelling. Migration tests typically are not applied. Material tests on preforms have the advantage that the concentration of migration relevant substances in PET is at least a factor of 1000 higher than in the concentration in food (simulants) after migration tests. Therefore, the applied analytical screening tests on preforms are much easier to validate and less expensive. In addition analysis of the PET preforms or bottles require no storage time for the migration test. Therefore the results are available within hours instead of ten days. Screening tests are therefore suitable for production control. The missing link, however, is the maximum concentration of NIAS in PET preforms or bottles which are corresponding to the 10 µg/l maximum (or any other) migration limit. In addition the migration is depending on the storage time, the bottle size, the storage temperature and the maximum bottle wall concentration. Therefore the maximum acceptable concentrations in PET vary depending on storage conditions and preform weight. Migration modelling is able to close this important gap.
Nonylphenol
The method of choice for PET preform or bottles testing towards migration relevant, non-intentionally added substances is headspace gas chromatography. The method applied at the Fraunhofer IVV was developed and validated within the European “Recyclability” Project in 2001. For identification of the determined NIAS headspace gas chromatography is coupled with mass spectrometry. As a result the identity as well as the approximate concentration is available from the screening test. Even without a mass spectrometer the molecular weight of migrating compounds can be predicted from their retention time. The molecular weight, or more precisely the molecular volume, can be used to predict the diffusion coefficient in PET.
Diethylhexyl phthalate
The diffusion coefficient can be considered as the most important factor for establishing a certain migration level in the PET packed beverages. For compliance evaluation it is important that migration modelling is overestimated. This means that the predicted migration should be in any case higher than the real migration into food under existing storage conditions. On the other hand, the migration should not be too overestimated otherwise the very low 10 µg/l limit is exceeded only due to overestimated migration calculation conditions. A realistic prediction model for the diffusion coefficients should be applied therefore. Such a prediction model has been developed by the Fraunhofer IVV (Welle 2013). The predicted diffusion coefficients are in perfect agreement with experimental data available from experimental migration kinetics. The model has been validated for temperatures between 20 °C and 100 °C. A scientific publication of the validation data is in progress. It has been shown that the overestimated character which is necessary for compliance evaluation can be established if the diffusion coefficients are assumed to be 20% higher than the diffusion coefficients derived from realistic prediction models.
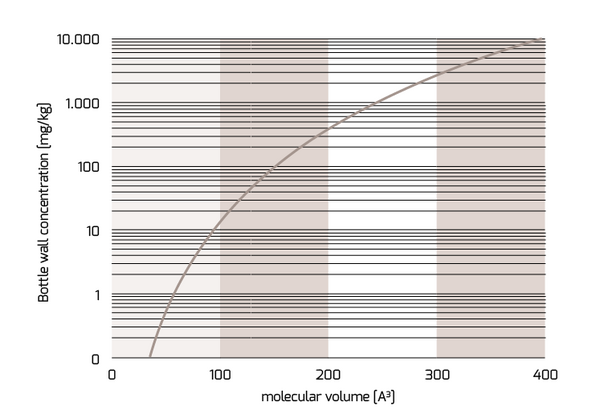
Fig. 1: Correlation between the bottle wall concentration and the molecular volume of NIAS corresponding with a migration of 10 µg/l after storage for 365 days at 25 °C (1 l in contact with 6 dm2)
Figure 1 shows the maximum bottle wall concentration depending on the molecular volume of the NIAS corresponding with the maximum migration level of 10 µg/l. The migration was calculated for a bottle volume of 1 l with an inner surface area of 600 cm2 for a storage time of 365 d at room temperature. The thickness of the bottle wall was 200 µm. Similar curves can be established for any other storage conditions and preform weights. The molecular volume of determined NIAS can be easily predicted from free internet programs. Some examples are given in Table 1.
The main conclusion of the Figure 1 is that the small molecules with low molecular volumes are the most important ones for PET compliance evaluation. NIAS with molecular volumes above 150 Å3 needed concentrations above of 100 mg/kg in the PET bottle wall in order to exceed the migration limit of 10 µg/l after storage for one year at room temperature. NIAS in the concentration ranges of 100 mg/kg in PET preforms are extremely rare and for shorter storage times as 365 days the maximum bottle wall concentration increases accordingly. Examples for molecules with molecular volume above 150 Å3 are typical substances of concern mentioned in public media like bisphenol A (221 Å3), nonylphenol (243 Å3), and diethylhexyl phthalate (411 Å3). The intentionally added substance anthranilamide has a molecular weight of 123 Å3. Anthranilamide, however, is approved for PET bottles for beverages up to a migration level of 50 µg/l. Due to the fact that the migration is directly proportional to the concentration in the PET bottle a concentration of 420 mg/kg is applicable for storage times of 365 days at room temperature. Acetaldehyde (47 Å3) should be in the concentration range of 1mg/kg otherwise the 10 µg/l migration limit might be exceeded which is coincidentally also the organoleptic threshold limit for acetaldehyde in natural mineral water.
In conclusion the following procedure can be applied for the evaluation of non-intentionally added substances in PET during preform production:
-screening of PET preforms or bottles as a production control by use of headspace GC
-determination of the concentration of any NIAS in the PET bottle
-prediction molecular volume of the determined NIAS
-comparison of the maximum concentrations in the PET bottle wall (according to the foreseeable contact conditions) with the actual determined concentration levels as pass/fail criteria
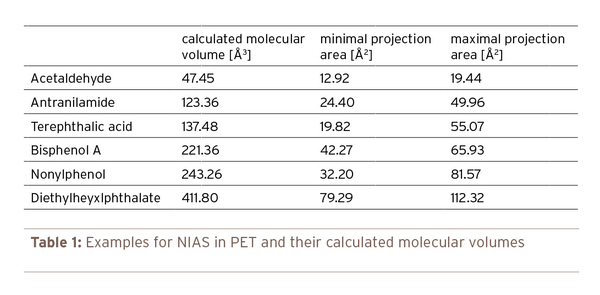
Table 1: Examples for NIAS in PET and their calculated molecular volumes
The method described within this study is an effective production control test for determination of any NIAS in PET preforms and bottles. The determination of NIAS at a detection limit of 10 µg/l is very time consuming and expensive. Therefore the 10 µg/l threshold limit has been translated into maximum concentration (QM) values within the PET bottles. The European Plastics Regulation 10/2011 gives such a flexibility. Also the use of scientifically recognized migration models is established in the Plastics Regulation for compliance confirmation.
The comPETence center provides your organisation with a dynamic, cost effective way to promote your products and services.

magazine
Find our premium articles, interviews, reports and more
in 3 issues in 2025.






